Chemical weathering creates landscape features through the breakdown of rocks and minerals by chemical reactions. Key features include caves, sinkholes, rounded boulders, and dissolved minerals in water. These processes occur due to water, carbonic acid, and atmospheric gases interacting with earth materials over long periods.
What Is Chemical Weathering?
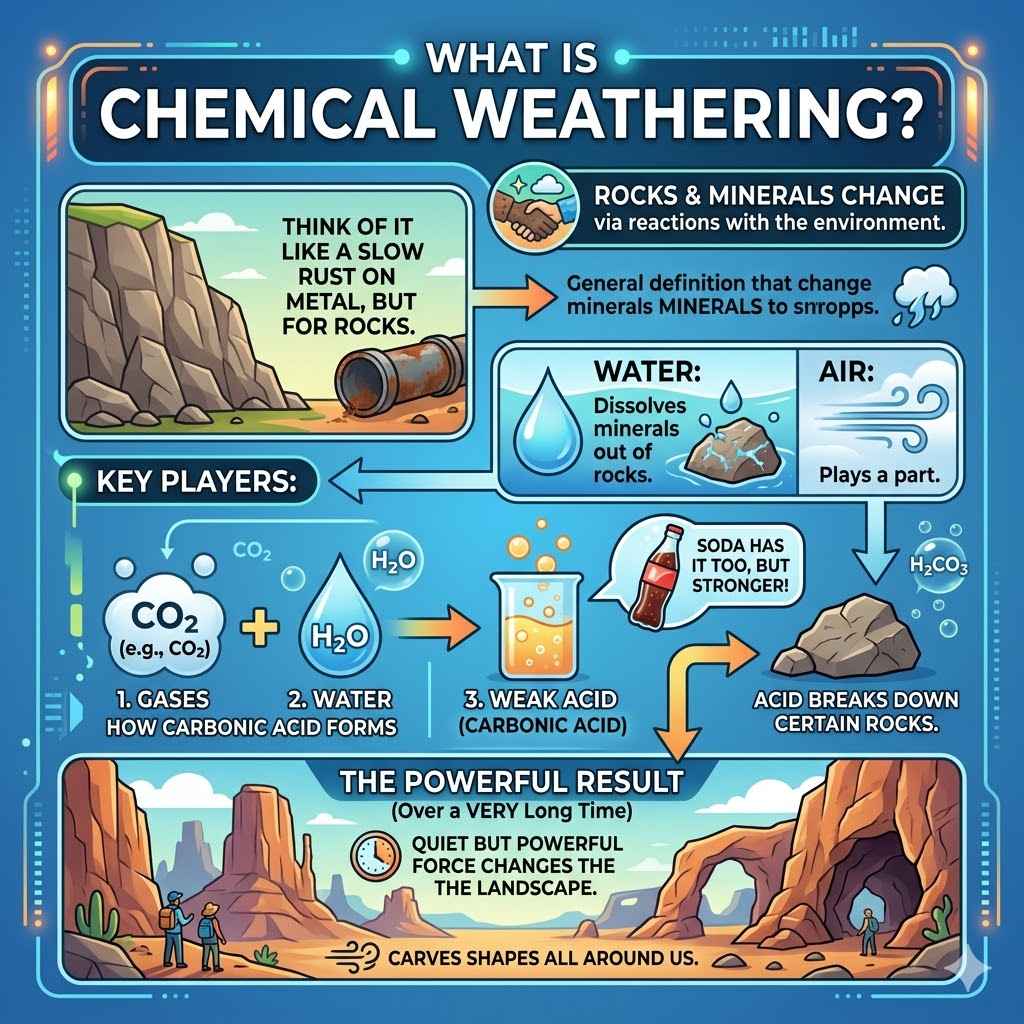
Chemical weathering is how rocks and minerals change. This happens when they react with things in the environment. Think of it like a slow rust on metal, but for rocks.
Water is a big player here. It can dissolve some minerals right out of a rock. Air also plays a part.
Gases like carbon dioxide can mix with water. This makes a weak acid.
This weak acid is called carbonic acid. It’s the same stuff in fizzy drinks, but much weaker. When this acid touches certain rocks, it can break them down.
Over a very, very long time, this process changes the landscape. It carves out shapes we see all around us. It’s a quiet force, but very powerful.
How Chemical Reactions Shape the Land
Several types of chemical reactions are at work. One is dissolution. This is when a mineral simply dissolves in water.
Think of salt disappearing in water. Some rocks, like limestone, are made of minerals that dissolve easily. Water flowing over limestone can slowly eat away at it.
Another reaction is oxidation. This is similar to rusting. When iron-rich minerals are exposed to oxygen and water, they change.
They form new minerals, like rust. This can make rocks weaker and change their color. You might see reddish or yellowish stains on rocks because of this.
Hydrolysis is also important. This is when water reacts with minerals. It changes the minerals into new ones.
These new minerals are often clay. Clay minerals are softer and weaker than the original minerals. This makes the rock easier to break apart.
Carbonation is a big one for limestone. Carbon dioxide from the air mixes with rainwater. This forms carbonic acid.
This acid then reacts with calcium carbonate, the main mineral in limestone. This reaction turns the solid limestone into soluble calcium bicarbonate. This then gets washed away by the water.
My First Encounter with Weathered Landscapes
I remember my first trip to Mammoth Cave National Park in Kentucky. I was maybe ten years old. The sheer size of the underground passages blew my mind.
Our guide explained how water had slowly carved out these massive rooms and tunnels. He talked about carbonic acid eating away at the limestone.
He pointed out stalactites hanging from the ceiling and stalagmites rising from the floor. He said these were formed by minerals dripping from the cave roof. As the water dripped, it left behind tiny bits of calcium carbonate.
It took thousands of years for them to grow. I touched the cool, damp cave wall. It felt smooth.
I could almost feel the water working its magic. It was then I truly understood that the earth was constantly changing, slowly and surely.
Key Chemical Weathering Agents
Water: The universal solvent. It dissolves many minerals and is essential for other reactions.
Carbon Dioxide: Mixes with water to form carbonic acid, a key agent in dissolving limestone.
Oxygen: Causes oxidation, changing iron-bearing minerals into rust.
Acids: Both natural (carbonic acid) and from pollution (acid rain) can break down rocks.
Living Organisms: Plant roots can release weak acids. Bacteria also produce chemicals that alter rocks.
Common Landscape Features Caused by Chemical Weathering
When these chemical reactions happen over vast amounts of time, they create specific shapes in the land. These are the features that make our planet so interesting to explore. We can see the evidence of this slow, steady work everywhere.
Caves and Karst Landscapes
Perhaps the most famous features of chemical weathering are caves. These are underground hollows. They form when water seeps into the ground.
If the ground is made of soluble rock like limestone, the water carries away bits of the rock. This is especially true if the water has carbonic acid in it.
Over thousands of years, the water creates passages. These passages can grow into large caves. The landscape above these caves is called a karst landscape.
Karst areas often have unique features. You might see sinkholes, disappearing streams, and underground rivers. It’s like the land is full of hidden secrets.
What is a Karst Landscape?
Definition: A region where the bedrock is soluble rock, usually limestone, and has been dissolved by water.
Key Features:
- Sinkholes: Depressions in the ground. They form when the roof of an underground cave collapses.
- Caves: Underground passages and chambers. Formed by dissolution.
- Disappearing Streams: Rivers that flow underground into sinkholes.
- Natural Bridges: Arches formed when a cave roof partially collapses.
- Uvalas and Poljes: Large, flat-bottomed depressions. They are like giant sinkholes or groups of sinkholes.
Sinkholes
Sinkholes are a direct result of chemical weathering. They happen when underground cavities in limestone or other soluble rocks are eroded. The ground above these cavities becomes weak.
Eventually, the roof of the cavity collapses. This creates a hole in the ground. Sometimes they are small.
Other times, they can be quite large and dangerous.
Many sinkholes are covered by soil and vegetation. You might not even see them until they suddenly appear. They are a clear sign that the earth below is being shaped by water and dissolving rock.
This is why areas with a lot of limestone are prone to sinkhole formation.
Rounded Boulders (Spheroidal Weathering)
Sometimes, large rocks, especially those made of granite, don’t break into sharp pieces. Instead, they become rounded. This is called spheroidal weathering.
It happens because the corners and edges of a rock are exposed to weathering more than the flat surfaces. Water can get into cracks more easily.
Chemical weathering attacks these exposed areas. It rounds off the rock. Imagine a cube of sugar left in water.
The edges will wear away faster than the flat sides. Over a long time, this makes the rock look like a ball or a smoothed-out shape. You often see these rounded boulders in fields or on hillsides.
Contrast: Spheroidal Weathering vs. Mechanical Weathering
Spheroidal Weathering (Chemical):
- Process: Chemical reactions attack cracks and edges more than faces.
- Result: Rounded boulders.
- Example: Granite boulders that look like smoothed stones.
Mechanical Weathering:
- Process: Physical forces break rocks into smaller pieces.
- Result: Angular, sharp-edged fragments.
- Example: Frost wedging creating sharp pieces of rock.
Rock Formations and Hoodoos
Unique rock formations are often shaped by chemical weathering. Take hoodoos, for example. These are tall, thin spires of rock.
They often have a capstone on top. Chemical weathering helps to carve away the softer rock around them. This leaves the harder parts standing.
Water can drip down the sides of a rock formation. If there are different types of rock, some will dissolve or weaken faster than others. This selective weathering carves out interesting shapes.
Wind and rain also play a role, but the chemical breakdown is key to creating these intricate forms.
Dissolved Load in Waterways
You can’t always see the effects of chemical weathering. But it’s happening in the water around us. Water flowing over rocks dissolves minerals.
This dissolved material is called the dissolved load. Rivers and streams carry these dissolved minerals to the ocean. This is a major way that rocks are broken down.
The chemistry of the water affects what gets dissolved. For example, water in areas with lots of limestone will have more dissolved calcium and bicarbonate. This is why some water sources are harder than others.
This constant dissolving is a quiet but significant part of the rock cycle.
Factors Influencing Chemical Weathering Rates
Not all rocks weather at the same speed. Several things make chemical weathering happen faster or slower. It’s a complex dance of chemistry and the environment.
Climate
Climate is a huge factor. Warm and wet climates speed up chemical weathering. Heat gives molecules more energy to react.
Water provides the medium for most chemical reactions. So, tropical rainforests are places where chemical weathering happens very quickly.
Cold and dry climates, on the other hand, slow it down. There’s less water to cause reactions. And the lower temperatures mean reactions happen more slowly.
This is why rocks in deserts or polar regions tend to last much longer before breaking down.
Rock Type
The type of rock makes a big difference. Some minerals are more stable than others. For example, quartz is very resistant to chemical weathering.
It’s why you find sand grains made of quartz after other minerals have worn away. Limestone, made of calcium carbonate, dissolves easily.
Minerals like feldspar can undergo hydrolysis. They turn into clay minerals. This weakens the rock.
Igneous rocks like granite have different minerals. Some, like mica and feldspar, weather chemically. Others, like quartz, are more resistant.
This leads to the characteristic rounded shapes of weathered granite.
Relative Stability of Minerals
- Most Stable (Resistant to Chemical Weathering): Quartz, iron oxides (like hematite).
- Moderately Stable: Feldspars (weather to clay), micas.
- Least Stable (Quickly Weathered): Olivine, pyroxene, amphibole, calcite (in limestone).
Think of it this way: Minerals that form at high temperatures (like in magma) tend to break down faster at the Earth’s surface.
Surface Area
The more surface a rock has exposed to the elements, the faster it will weather. This is why rocks that are broken into many small pieces weather much faster than one big solid rock. Think about crushing a pill versus swallowing it whole.
The crushed pill dissolves faster.
Cracks and fractures in rocks increase the surface area. Water can seep into these cracks. This allows chemical reactions to start from within the rock.
This is why spheroidal weathering can happen where there are initial cracks. It’s the edges and corners that get attacked first.
Presence of Acids
The amount and type of acid present greatly affect weathering rates. Carbonic acid, formed from carbon dioxide and water, is common. But other acids can be present.
For instance, acid rain, caused by pollution, can significantly speed up the weathering of certain rocks, like limestone and marble.
Even weak organic acids released by plant roots can contribute to weathering. As roots grow in cracks, they can release acids that slowly break down the rock. This is a small-scale effect, but over time, it adds up.
Real-World Examples and Scenarios
Seeing these features in real life makes the concept much clearer. It’s not just something in textbooks. It’s shaping the world we live in.
Visiting a Limestone Region
If you travel to areas known for limestone, like parts of Florida, the Yucatan Peninsula, or southern Indiana, you’ll see many karst features. You’ll notice the lack of surface rivers. The water seems to vanish.
You’ll see the numerous sinkholes dotting the landscape.
In these regions, houses and roads can be built on top of underground cave systems. When a cave roof collapses, it can cause serious damage. This is a direct human impact from natural chemical weathering processes.
Examining Old Buildings and Statues
Think about old marble statues or buildings made of limestone. Over time, their details become worn and rounded. This is a visible example of chemical weathering in action.
The softest parts of the stone dissolve or break down faster.
Statues that were once sharp and clear can become smooth and indistinct. Acid rain has been a major culprit in speeding up this process in cities. It shows how even man-made structures are subject to the slow power of nature’s chemistry.
Exploring Caves
Going into a cave is like stepping into a laboratory of chemical weathering. You see stalactites and stalagmites. These are formed by the slow dripping of mineral-rich water.
Each drop leaves behind a tiny deposit. It takes thousands of years to build these formations.
The walls of the cave are themselves a testament to dissolution. They show the pathways water took. They demonstrate how entire underground systems can be carved out.
The smooth, often wavy surfaces you see are the result of this constant erosion by water.
Quick Scan: Signs of Chemical Weathering
Look for:
- Smooth, rounded rocks in areas with soluble rock.
- Holes or depressions in the ground (sinkholes).
- Underground streams or disappearing rivers.
- Caves and karst topography.
- Worn-down details on old stone buildings or statues.
- Stalactites and stalagmites in caves.
- Reddish or yellowish stains on rocks (oxidation).
What This Means for You
Understanding chemical weathering helps us appreciate the dynamic nature of our planet. It also has practical implications.
When Is It Normal?
It is completely normal for rocks and landscapes to change due to chemical weathering. This is a natural, ongoing process. Karst landscapes with sinkholes are normal in areas with limestone.
Rounded boulders are also a common sight in many regions.
The slow erosion of coastlines, the dissolution of minerals in your tap water, and the formation of caves are all part of this natural cycle. It’s how the earth shapes itself over geological time.
When Should You Be Concerned?
You should be concerned when chemical weathering happens rapidly or causes immediate danger. Rapid sinkhole formation, especially near homes or infrastructure, is a major concern. This can happen when underground water flow changes, or when heavy construction alters the ground.
Severe acid rain can accelerate the decay of buildings, bridges, and natural rock formations much faster than normal. This damages historical sites and can weaken structures. If you live in a karst region, being aware of potential sinkhole risks is important for safety.
Simple Checks and Observations
If you live in a limestone-rich area, keep an eye out for new cracks in the ground or subtle depressions. If you notice unusually fast wear on any stone features around your home, it might be worth investigating. Sometimes, changes in water quality could also hint at increased mineral dissolution.
Taking note of your surroundings can help you spot potential issues early. It’s about being aware of the slow, powerful forces that shape the land beneath our feet. You don’t need to be a geologist to notice these things.
Quick Fixes and Tips
While you can’t stop natural chemical weathering, you can manage some of its impacts.
For Homeowners in Karst Regions
If you live in an area prone to sinkholes, it’s wise to consult with local geologists or engineers. They can assess your property for risks. Proper drainage is key; avoid concentrating water flow in one area.
Be cautious with any new construction that might alter underground water patterns.
If you see a small depression forming, report it to your local authorities. Early detection can sometimes prevent larger problems.
Protecting Stone Structures
For historical buildings or statues, protective coatings can sometimes be applied. These coatings act as a barrier against acid rain and water. However, these solutions must be carefully chosen to avoid damaging the stone itself.
Professional advice is often needed.
Ensuring good drainage around stone structures is also important. Water pooling can accelerate weathering.
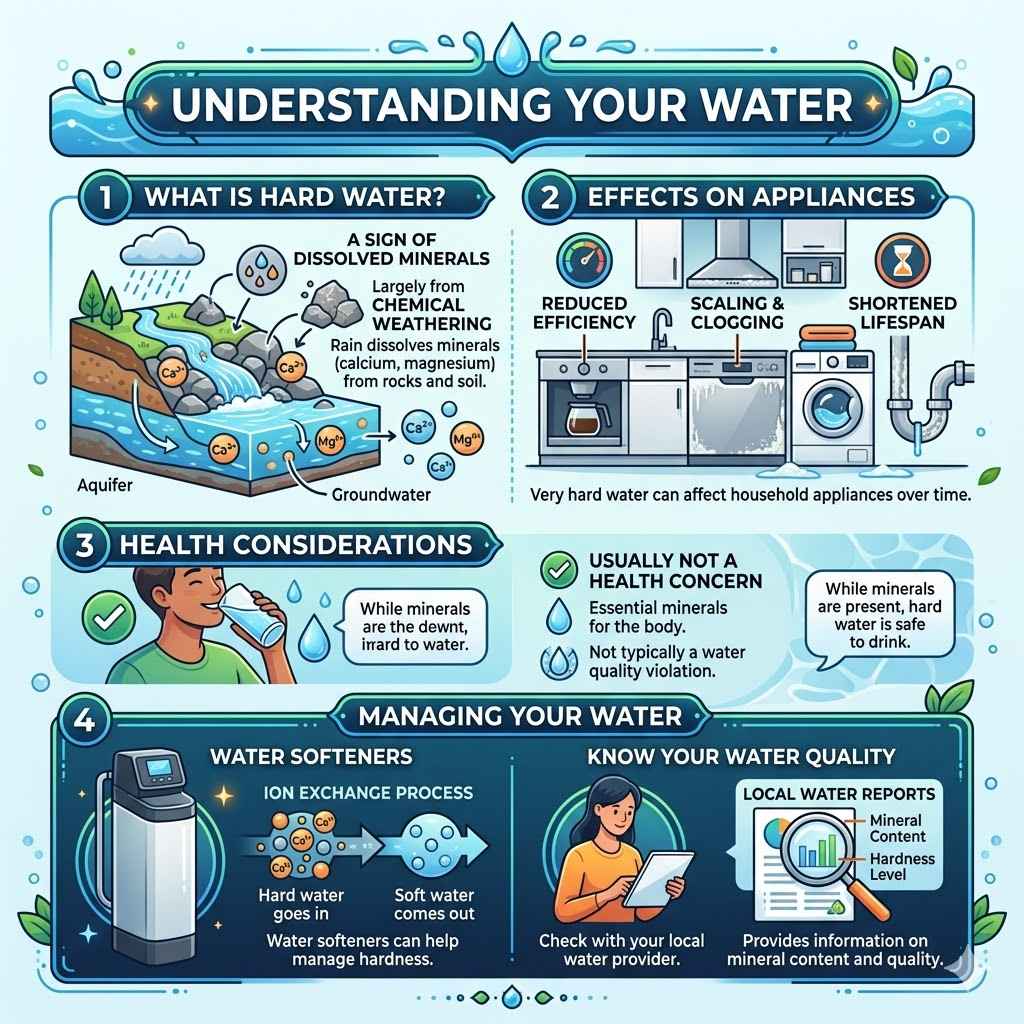
Understanding Your Water
If you have hard water, it’s a sign of dissolved minerals, largely from chemical weathering. While usually not a health concern, very hard water can affect appliances. Water softeners can help manage this.
In areas with significant mineral dissolution, it’s good to know what’s in your water. Local water reports often provide this information.
Frequently Asked Questions
What is the difference between chemical weathering and physical weathering?
Physical weathering breaks rocks into smaller pieces without changing their chemical makeup. Examples include frost wedging and abrasion. Chemical weathering changes the rock’s composition through reactions with water, gases, or acids.
This breaks down the minerals that make up the rock.
Which rocks are most affected by chemical weathering?
Rocks made of minerals that dissolve easily are most affected. Limestone (calcium carbonate) is a prime example. Others include dolomite, gypsum, and salt beds.
Rocks rich in iron-bearing minerals are also prone to oxidation.
How long does it take for chemical weathering to form a cave?
It takes a very long time, often thousands to hundreds of thousands of years. The process depends on the rock type, the amount of rainfall, the acidity of the water, and the rate of water flow through the rock.
Can acid rain cause landscape features?
Yes, acid rain can significantly speed up chemical weathering. It can cause statues and buildings made of limestone or marble to erode much faster than normal. In natural settings, it can accelerate the breakdown of soluble rocks and contribute to features like rapid cave formation or erosion.
Are rounded boulders always a sign of chemical weathering?
Rounded boulders are a strong indicator of spheroidal weathering, which is a type of chemical weathering. However, some rounding can occur through mechanical processes like abrasion over very long periods. But the distinct smooth, spherical shapes are usually due to chemical attack on the rock’s weaknesses.
How does chemical weathering contribute to soil formation?
Chemical weathering breaks down the parent rock into smaller mineral particles. These particles are then mixed with organic matter from dead plants and animals. This mixture forms soil.
Chemical reactions also alter some minerals into new ones, like clays, which are important components of soil.
Conclusion
Chemical weathering is a fascinating and powerful force. It’s always working, slowly shaping the earth’s surface. From vast cave systems to the rounded rocks in a field, these features are a testament to its quiet work.
Understanding these processes helps us appreciate the dynamic planet we live on.






